 I believe in evidence-based decision making. Our RRPs are smoke-free products that produce an aerosol that contains far lower quantities of harmful and potentially harmful constituents than found in cigarette smoke. We call it: Examples Level of importance for you to have multiple examples or exposures to something in order to be convinced about it. PMI has a range of RRPs in various stages of development, scientific assessment and commercialization. Evidence based decision making Introduction Health policy in the broadest sense can be defined as those actions of governments and other actors in the society. 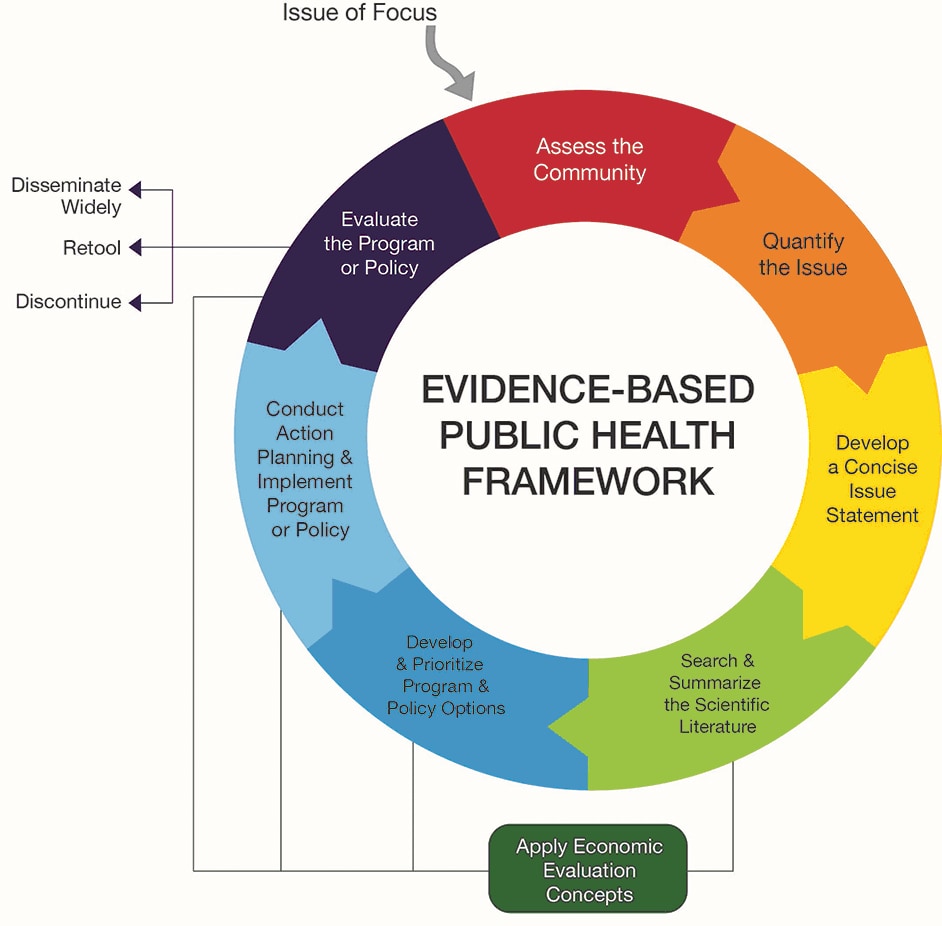 
Reduced Risk Products ("RRPs”) is the term we use to refer to products that present, are likely to present, or have the potential to present less risk of harm to smokers who switch to these products versus continuing smoking. The purpose of this site is not for advertising or marketing. Evidence-based policy is a concept in public policy that advocates for policy decisions to be grounded on, or influenced by, rigorously established. Put simply, it means finding, using and sharing what works in public health.’. This site is for use by scientists, the public health and regulatory communities, and other stakeholders with an interest in tobacco policy. Evidence-informed decision-making (EIDM) is ‘the process of distilling and disseminating the best available evidence from research, practice and experience and using that evidence to inform and improve public health policy and practice. Inherent in these definitions is the recognition that research evidence is a valued component of the clinical decision-making process, and the intent is that the use of current best evidence does not replace clinical skills, judgment, or experience but provides another dimension to the decision-making process that also considers the patient’s preferences (Figure 1). is operated by Philip Morris International for the purpose of publishing and disseminating scientific information about Philip Morris International’s efforts to develop and assess products that have the potential to reduce individual risk and population harm associated with tobacco use. EBDM is distinguished from the use of evidence-based practices (EBP), which is the application of specific research findings to discrete practices. For example, the American Dental Association (ADA) defines "evidence-based dentistry" (EBD) as: an approach to oral health care that requires the judicious integration of systematic assessments of clinically relevant scientific evidence, relating to patient’s oral and medical condition and history, with the dentists’ clinical expertise and the patient’s treatment needs and preferences. Evidence-based decision making (EBDM) is the practice of using research to inform decisions throughout the justice system. Several professions have adapted this definition to make it specific to their discipline. Rather than refer to medicine, often this definition has been broadened to mean ‘practice’ or ‘healthcare’ and is the definition we are using for Evidence-Based Practice (EBP). The integration of the best research evidence with our clinical expertise and our patient's unique values and circumstances. This new methodology was termed Evidence-Based Medicine (EBM) 1 and its definition is currently stated as: The old clinical problem-solving model based on individual experience or the use of information gained by consulting authorities (colleagues or text books) gave way to a new methodology for practice and restructured the way in which more effective clinical problem-solving should be conducted. PDF Evidence-based practice (EBP) is an orientation to practice that values evidence as a resource for clinical decision making while recognizing that. As clinical research and the publication of findings increased, so did the need to use the medical literature to guide practice. about the quality of decisions in government have prompted an increased focus on data-driven or evidence- based decision making practices. Consequently, various fields require their practitioners to have an in-depth understanding of judgment and decision-making. Using evidence from the medical literature to answer questions, direct clinical action and guide practice was pioneered at McMaster University, Ontario, Canada in the 1980’s. Because we rely on well-designed research studies to demonstrate the efficacy and effectiveness of diagnostic tests, treatment strategies, new materials, and products, knowing how to find the scientific evidence is an essential component for clinical practice. 
Evidence has always contributed to clinical decision-making however, with the proliferation of clinical studies and journal publications, keeping current with relevant research is nearly impossible. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
